SARS-CoV-2 Neutralizing Antibody Rapid Test Device
KIT COMPONENTS
| Individually packed test devices | Each device contains a strip with colored conjugates and reactive reagents pre-spreaded at the corresponding regions |
| Disposable pipettes | For adding specimens use |
| Buffer | Phosphate buffered saline and preservative |
| Package insert | For operation instruction |
ASSAY PROCEDURE
Ring the specimen and test components to room temperature Mix the specimen well prior to assay once thawed. Place the test device on a clean, flat surface.
For capillary whole blood sample:
To use a capillary tube: Fill the capillary tube and transfer approximately 25 µL (or 1 drop) of fingerstick whole blood specimen to the specimen well (S) of the test device, then add 1 drop (about 30 µL) of Sample Diluent immediately into the sample well.
For whole blood sample:
Fill the dropper with the specimen then transfer 1 drop (about 25 µL) of specimen into the sample well. Making sure that there are no air bubbles. Then transfer 1drop (about 30 µL) of Sample Diluent immediately into the sample well.
For Plasma/ Serum sample:
Fill the dropper with the specimen then transfer 25 µL of specimen into the sample well. Making sure that there are no air bubbles. Then transfer 1 drop (about 30 µL) of Sample Diluent immediately into the sample well.
Set up a timer. Read the result at 10 minutes. Don’t read result after 20 minutes.
To avoid confusion, discard the test device after interpreting the result.
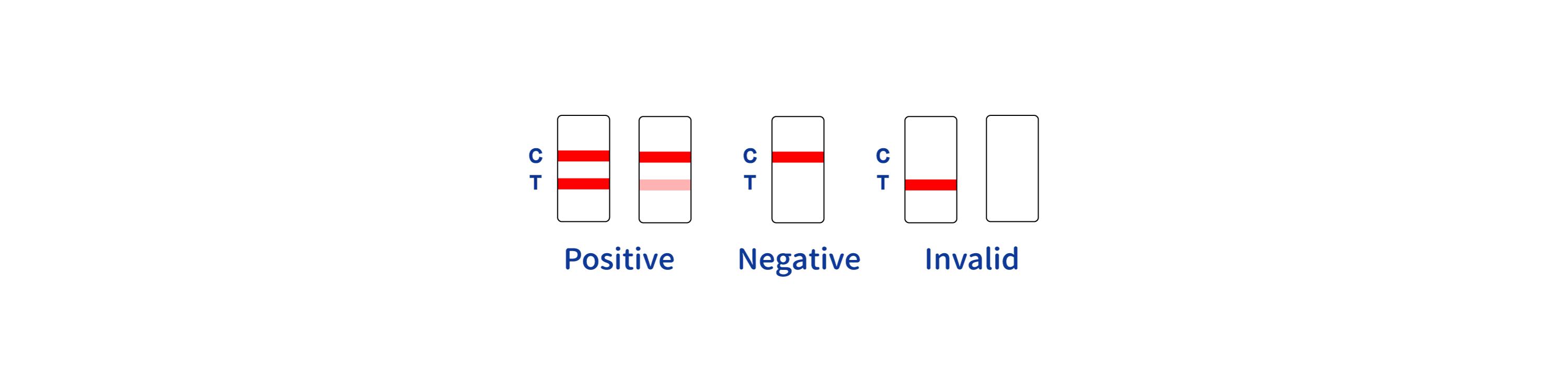
ASSAY RESULT
PRECAUTIONS
1.For professional in vitro diagnostic use only.
2.Do not use after the expiration date indicated on the package. Do not use the test if the foil pouch is damaged. Do not reuse tests.
3.The extraction reagent solution contains a salt solution if the solution contacts the skin or eye, flush with copious amounts of water.
4.Avoid cross-contamination of specimens by using a new specimen collection container for each specimen obtained.
5.Read the entire procedure carefully prior to testing.
6.Do not eat, drink or smoke in the area where the specimens and kits are The SARS-CoV-2 Neutralizing Antibody Rapid Test Cassette has been tested for anti-influenza A virus, anti-influenza B virus, anti-RSV, anti-Adenovirus, HBsAb, anti-Syphilis, anti-H. Pylori, anti-HIV, anti-HCV and HAMA positive specimens. The results showed no cross-reactivity.