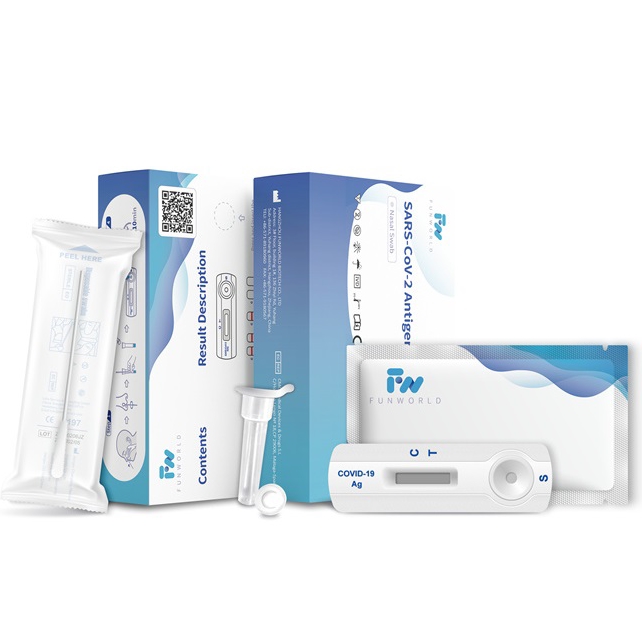
Recently, SARS-Cov-2 Antigen Rapid Test Device (Self-Testing) independently developed by Hangzhou Funworld Biological Co., Ltd. successfully obtained the EU CE self-test certificate.The approval of this certification means that SARS-Cov-2 Antigen Rapid Test Device (Self-Testing)developed by Funworld Biotech can be marketed in the 27 EU member states and other countries that recognize the EU CE certification. Consumers can purchase in supermarkets, pharmacies and other channels and conduct simple and rapid self-testing.
In addition, the SARS-Cov-2 Antigen Rapid Test Device (Self-Testing) produced by Funworld Biotech have also been included in the EU general white list HSC Common List. Funworld Bio’s antigen test products have been unanimously recognized by distributors and users for their excellent performance and excellent quality. The CE self-test certification of the new crown antigen self-test product further improves the company’s product line, enhances the company’s new crown detection product’s international competitiveness, and lays a solid foundation for the steady development of the new crown detection business in the era of normalized epidemic prevention and control .
Since establishment, Funworld Bio has been focusing on the field of POCT (point-of-care testing), mainly engaged in the research and development, production and sales of POCT diagnostic reagents.Existing products cover five major testing fields, including drug abuse (drug) tests, infectious disease tests, tumor marker tests, and Cardiac marker tests and fertility test kits.
Post time:May-31-2022
Post time: 2023-11-20 16:51:08